
How Small Molecules Create Big Medical Breakthroughs
In the hidden world of chemistry, tiny molecular structures quietly influence life and health. A single carefully designed molecule can alter how diseases develop, spread, or respond to treatment. Some molecules slow down cancer growth, others protect brain cells, and some prevent viruses from replicating.
Today, researchers across the world are working to develop innovative therapeutic molecules that could transform modern medicine. While many of these compounds are still under investigation, their chemical mechanisms show remarkable promise. This article highlights five powerful molecules that may redefine how major diseases are treated.
Blarcamesine: A Potential Breakthrough for Neurodegenerative Diseases
Blarcamesine is a small organic heterocyclic molecule known for its interaction with neurological receptors. Its structure, consisting of aromatic rings and functional groups, allows it to bind effectively to receptor sites in nerve cells.
Chemical Mechanism of Action
Blarcamesine primarily acts as a sigma-1 receptor agonist. Sigma-1 receptors are located in the endoplasmic reticulum and play a critical role in regulating cellular stress and calcium signaling.
When blarcamesine binds to these receptors, it helps stabilize protein folding and reduces oxidative stress in neurons. The interaction involves hydrogen bonding, hydrophobic interactions, and π–π stacking, which strengthen receptor binding.
Disease Targeted
• Alzheimer’s disease
• Parkinson’s disease
• Rett syndrome
Stage of Research
Blarcamesine is currently undergoing advanced clinical trials for neurodegenerative disorders.
Chemical Significance
This molecule demonstrates how ligand-receptor interactions can protect neurons by regulating intracellular signaling pathways.
Zelenirstat: Enzyme Inhibition Through Molecular Design
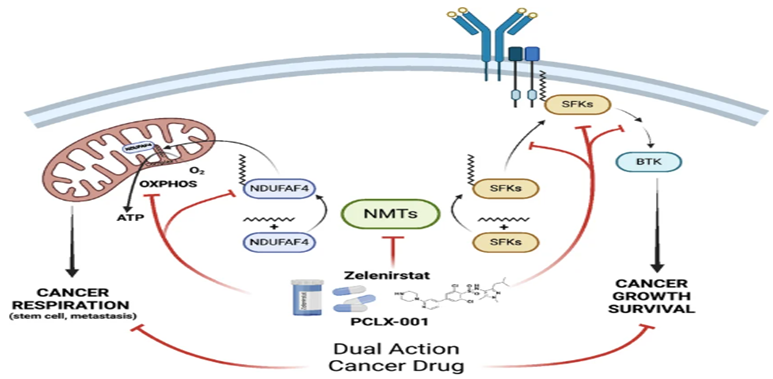
Zelenirstat, also known as PCLX-001, is a small molecule inhibitor targeting N-myristoyltransferase (NMT), an enzyme essential for protein modification.
Mechanism of Action
NMT enzymes catalyze myristoylation, a process where fatty acids are attached to proteins, influencing their function and localization.
Zelenirstat mimics the natural substrate of the enzyme and blocks its active site, preventing the transfer of myristic acid. This inhibition disrupts cellular processes essential for cancer cell survival.
This includes:
• Competitive binding
• Non-covalent stabilization within the enzyme pocket
• Disruption of metabolic pathways
Disease Targeted
• Leukemia
• Solid tumors
• Viral infections dependent on lipid-modified proteins
Chemical Importance
Zelenirstat highlights the power of structure-based drug design in selectively targeting cancer cells.
Thapsigargin: A Natural Compound with Strong Biological Activity
Thapsigargin is a naturally derived compound obtained from plants of the Thapsia genus. It belongs to the sesquiterpene lactone class of molecules.
Chemical Structure
The molecule contains:
• A lactone ring
• Multiple oxygen-containing functional groups
• A rigid terpenoid backbone
Mechanism of Action
Thapsigargin inhibits the SERCA pump, which regulates calcium transport within cells. By binding to the transmembrane region, it blocks calcium movement and disrupts cellular balance.
This leads to:
• Calcium accumulation in the cytoplasm
• Endoplasmic reticulum stress
• Activation of programmed cell death
Disease Targeted
• Prostate cancer
• Brain tumors
• Other solid tumors
Chemical Innovation
Scientists have modified thapsigargin into prodrug forms that activate only within tumor cells, demonstrating targeted drug delivery.
ABBV-CLS-484: Controlling Immune System Chemistry
ABBV-CLS-484 is a synthetic small molecule designed to regulate immune signaling pathways, particularly in cancer treatment.
Chemical Properties
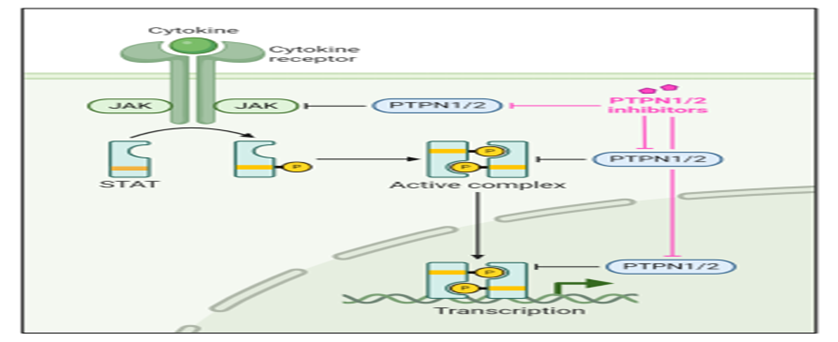
It is a protein tyrosine phosphatase inhibitor that targets key regulatory enzymes in immune cells.
Mechanism of Action
The molecule inhibits:
• PTPN1
• PTPN2
These enzymes normally remove phosphate groups from signaling proteins. By inhibiting them, the molecule enhances phosphorylation levels, leading to stronger immune responses.
This results in increased activity of T-cells and natural killer cells, improving the body’s ability to fight cancer.
Disease Targeted
• Cancers resistant to conventional immunotherapy
Chemical Impact
This compound demonstrates how modifying enzyme-driven signaling pathways can boost immune responses against tumors.
Nelfinavir: Drug Repurposing in Modern Medicine
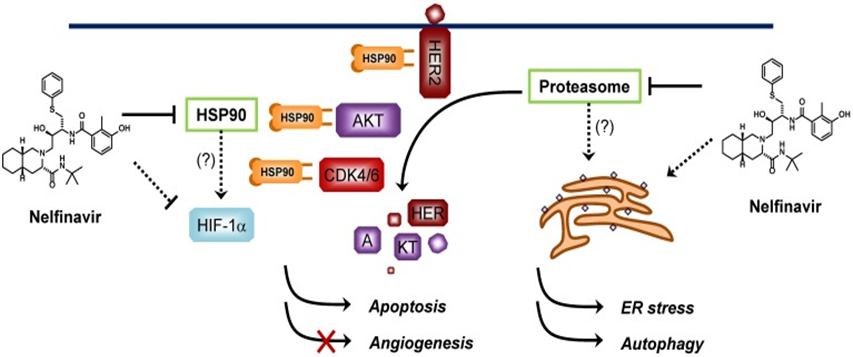
Nelfinavir is a well-known drug originally developed to treat HIV infections. It is now being explored for its potential in cancer therapy.
Chemical Structure
It contains functional groups such as:
• Amides
• Hydroxyl groups
• Aromatic rings
These features allow it to bind effectively to enzyme active sites.
Mechanism of Action
Nelfinavir inhibits HIV protease, preventing viral replication. Additionally, it affects cellular stress pathways and Akt signaling, which are crucial for cancer cell survival.
Disease Targeted
• HIV infection
• Brain tumors
• Prostate cancer
Chemical Significance
Nelfinavir represents the importance of drug repurposing, where existing medicines are used for new therapeutic applications.
Conclusion: The Future of Medicine Lies in Molecular Design
Modern medicine is increasingly shaped by molecular level innovations. The five molecules discussed here represent different strategies in medicinal chemistry, including receptor targeting, enzyme inhibition, calcium regulation, immune modulation, and drug repurposing.
Each of these compounds works through precise chemical interactions with biological systems. These interactions form the foundation of next-generation therapies.
As advancements in synthetic chemistry, computational modeling, and biotechnology continue, the ability to design highly selective and effective drugs will improve. In the future, many diseases that are currently difficult to treat may become manageable through carefully engineered molecules.
These discoveries remind us that even the smallest molecular structures can have a powerful impact on human health.
References
Maurice, T. (2025). Prevention of memory impairment and hippocampal injury with blarcamesine in an Alzheimer’s disease model. Neuroscience Letters, 138349.
Feldman, J. (n.d.). Phase 1/2 trial of oral zelenirstat launches in relapsed/refractory AML.
Jaskulska, A., Janecka, A. E., & Gach-Janczak, K. (2020). Thapsigargin from traditional medicine to anticancer drug. International Journal of Molecular Sciences, 22(1), 4.
Editor: Ayesha Noor
